
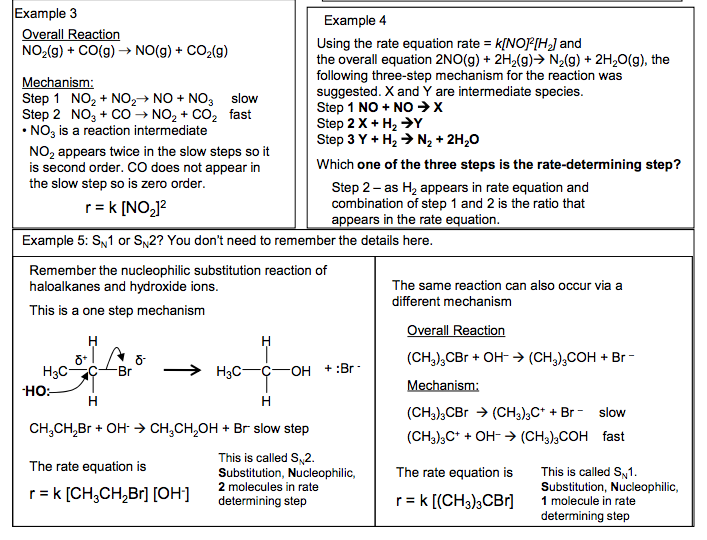
Rate Equations and Mechanisms A mechanism is a series of steps through which the reaction progresses, often forming intermediate compounds. If all the steps are added together they will add up to the overall equation for the reaction Each step can have a different rate of reaction. The slowest step will control the overall rate of reaction. The slowest step is called the rate-determining step. The molecularity (number of moles of each substance) of the molecules in the slowest step will be the same as the order of reaction for each substance. e.g. 0 moles of A in slow step would mean A is zero order. 1 mole of A in the slow step would mean A is first order Example 1 overall reaction A + 2B + C D + E Mechanism Step 1 A + B X + D slow Step 2 X + C Y fast Step 3 Y + B E fast r = k [A]1 [B]1 [C]o r = k [X]1 [C]1 The intermediate X is not one of the reactants so must be replaced with the substances that make up the intermediate in a previous step A + B X + D r = k[A]1 [B]1 [C]1 overall reaction A + 2B + C D + E Mechanism Step 1 A + B X + D fast Step 2 X + C Y slow Step 3 Y + B E fast Example 2 Example 3 Overall Reaction NO2 (g) + CO(g) NO(g) + CO2 (g) Mechanism: Step 1 NO2 + NO2 NO + NO3 slow Step 2 NO3 + CO NO2 + CO2 fast • NO3 is a reaction intermediate r = k [NO2 ]2 Example 5: SN1 or SN2? You don’t need to remember the details here. Remember the nucleophilic substitution reaction of haloalkanes and hydroxide ions. This is a one step mechanism CH3CH2Br + OH- CH3CH2OH + Br- slow step The rate equation is r = k [CH3CH2Br] [OH-] The same reaction can also occur via a different mechanism Overall Reaction (CH3 )3CBr + OH– (CH3 )3COH + Br – Mechanism: (CH3 )3CBr (CH3 )3C+ + Br – slow (CH3 )3C+ + OH– (CH3 )3COH fast The rate equation is r = k [(CH3 )3CBr] This is called SN2. Substitution, Nucleophilic, 2 molecules in rate determining step This is called SN1. Substitution, Nucleophilic, 1 molecule in rate determining step C is zero order as it appears in the mechanism in a fast step after the slow step NO2 appears twice in the slow steps so it is second order. CO does not appear in the slow step so is zero order. Using the rate equation rate = k[NO]2 [H2 ] and the overall equation 2NO(g) + 2H2 (g) N2 (g) + 2H2O(g), the following three-step mechanism for the reaction was suggested. X and Y are intermediate species. Step 1 NO + NO X Step 2 X + H2 Y Step 3 Y + H2 N2 + 2H2O Which one of the three steps is the rate-determining step? Example 4 Step 2 – as H2 appears in rate equation and combination of step 1 and 2 is the ratio that appears in the rate equation.
/
~
~
~
/
3.1.9.2 Determination of rate equation (A-level only)
The orders with respect to reactants can provide information about the mechanism of a reaction.
Students should be able to:
use the orders with respect to reactants to provide information about the rate determining/limiting step of a reaction.


 1.9 Rate Equations Page 7
1.9 Rate Equations Page 7 Oxford Textbook Pages : 293 - 295
Oxford Textbook Pages : 293 - 295 CGP Revision Guide Pages : 72 - 73
CGP Revision Guide Pages : 72 - 73