
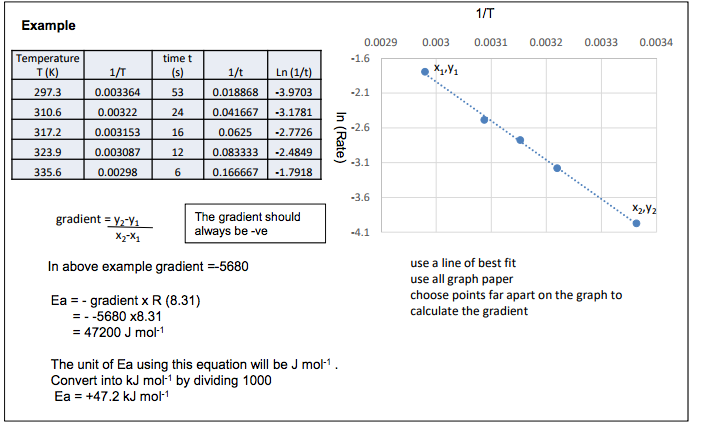
Increasing temperature increases the rate constant k. The relationship is given by the Arrhenius equation k = Ae-Ea/RT where A is a constant R is gas constant and Ea is activation energy. Increasing the temperature increases the value of the rate constant k The Arrhenius equation can be rearranged ln k = constant – Ea/(RT) k is proportional to the rate of reaction so ln k can be replaced by ln(rate) From plotting a graph of ln(rate) or ln k against 1/T the activation energy can be calculated from measuring the gradient of the line ln (Rate) 1/T Gradient = – Ea/ R Ea = – gradient x R Effect of Temperature on Rate Constant: The Arrhenius Equation -4.1 -3.6 -3.1 -2.6 -2.1 -1.6 0.0029 0.003 0.0031 0.0032 0.0033 0.0034 ln (Rate) 1/T use a line of best fit use all graph paper choose points far apart on the graph to calculate the gradient
/
~
~
~
/
The rate constant k varies with temperature as shown by the equation:
k = Ae–Ea/RT
where A is a constant, known as the Arrhenius constant, Ea is the activation energy and T is the temperature in K.
Students should be able to:
• explain the qualitative effect of changes in temperature on the rate constant k
• perform calculations using the equation k =Ae–Ea/RT
• understand that the equation k =Ae–Ea/RT can be rearranged into the form ln k = –Ea /RT + ln A and know how to use this rearranged equation with experimental data to plot a straight line graph with slope –Ea /R These equations and the gas constant, R, will be given when required.


 1.9 Rate Equations Page 6
1.9 Rate Equations Page 6 Oxford Textbook Pages : 287 - 292
Oxford Textbook Pages : 287 - 292 CGP Revision Guide Pages : 70 - 71
CGP Revision Guide Pages : 70 - 71





