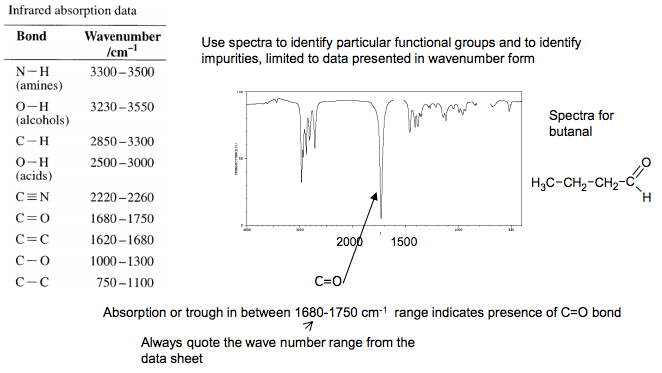

Infrared spectroscopy Certain groups in a molecule absorb infra-red radiation at characteristic frequencies Complicated spectra can be obtained than provide information about the types of bonds present in a molecule Below 1500 cm-1 – “Fingerprinting” Complicated and contains many signals – picking out functional group signals difficult. This part of the spectrum is unique for every compound, and so can be used as a “fingerprint”. A computer will compare the IR spectra against a database of known pure compounds to identify the compound Above 1500 cm-1 – “Functional group identification” Use an IR absorption table provided in exam to deduce presence or absence of particular bonds or functional groups e.g. C=O 1680 – 1750 cm-1 O-H (acid) 2500- 3000 cm-1 3.6 Analysis Carbon has a value of 12.0000 as it is by definintion the standard reference 1 Mass spectrum for butane 43 29 N Goalby chemrevise.org 2000 1500 Use spectra to identify particular functional groups and to identify impurities, limited to data presented in wavenumber form C=O Spectra for butanal “rogue” absorptions can also occur and are indicators of impurities O-H absorptions tend to be broad Spectra for ethanoic acid C=O The absorption of infra-red radiation by bonds in this type of spectroscopy is the same absorption that bonds in CO2 , methane and water vapour in the atmosphere do that maybe causing global global warming. Absorption or trough in between 1680-1750 cm-1 range indicates presence of C=O bond Absorption or trough in between 2500-3000 cm-1 range indicates presence of O-H bond in an acid Always quote the wave number range from the data sheet Absorption or trough in between 1680- 1750 cm-1 range indicates presence of C=O bond
/
~
~
~
/
3.3.6.3 Infrared spectroscopy
Bonds in a molecule absorb infrared radiation at characteristic wavenumbers.
‘Fingerprinting’ allows identification of a molecule by comparison of spectra.
Students should be able to:
• use infrared spectra and the Chemistry Data Sheet or Booklet to identify particular bonds, and therefore functional groups, and also to identify impurities. The link between absorption of infrared radiation by bonds in CO2 , methane and water vapour and global warming.


 3.6 Organic Analysis1 Page 2
3.6 Organic Analysis1 Page 2 Oxford Textbook Pages : 245 - 249
Oxford Textbook Pages : 245 - 249 CGP Revision Guide Pages : 158 - 159
CGP Revision Guide Pages : 158 - 159