
High resolution mass spectrometry can be used to determine the molecular formula of a compound from the accurate mass of the molecular ion Mass Spectrometry The peak with the highest mass/charge ratio will be normally due to the original molecule that hasn’t fragmented (called the molecular ion) . As the charge of the ion is +1 the mass/ charge ratio is equal to Mr. When organic molecules are passed through a mass spectrometer, it detects both the whole molecule and fragments of the molecule C4H10 = 58 Molecular ion formed: M → [M]+. + e– Definition: molecular ion is the molecule with one electron knocked off (It is both an ion and a free radical) High resolution mass spectroscopy can measure the mass to 5 d.p. This can help differentiate between compounds that appear to have similar Mr (to the nearest whole number) Accurate masses of atoms: H = 1.0078 C = 12.0000 O = 15.9949 N = 14.0031 For example, the following molecular formulas all have a rough Mr of 60, but a more precise Mr can give the molecular formula. e.g. Mr = 60.02112 molecular formula = C2H4O2 Mr = 60.05751 molecular formula = C3H8O Mr = 60.03235 molecular formula = CH4N2O Example 1 A compound is found to have an accurate relative formula mass of 46.0417. It is thought to be either CH3CH2OH or H2NCH2NH2 . Calculate the Mr of each compound to 4 decimal places to work out which one it is. CH3CH2OH = (12.0000 x 2) + (15.9949 x 1) + (1.0078 x6) = 46.0417 H2NCH2NH2 . = (12.0000 x 1) + (14.0031 x 2) + (1.0078 x6) = 46.0530
3.3.6.2 Mass spectrometry
Mass spectrometry can be used to determine the molecular formula of a compound.
Students should be able to use precise atomic masses and the precise molecular mass to determine the molecular formula of a compound.

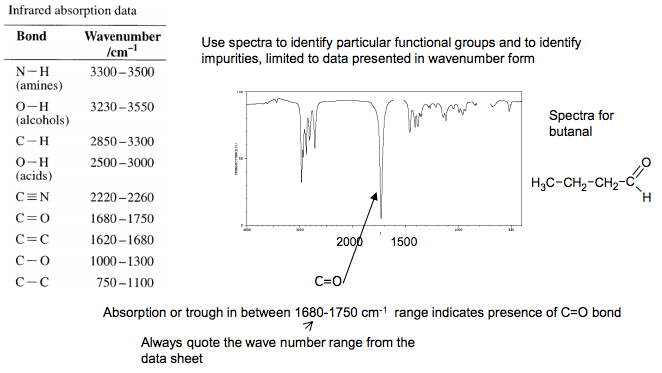

Infrared spectroscopy Certain groups in a molecule absorb infra-red radiation at characteristic frequencies Complicated spectra can be obtained than provide information about the types of bonds present in a molecule Below 1500 cm-1 – “Fingerprinting” Complicated and contains many signals – picking out functional group signals difficult. This part of the spectrum is unique for every compound, and so can be used as a “fingerprint”. A computer will compare the IR spectra against a database of known pure compounds to identify the compound Above 1500 cm-1 – “Functional group identification” Use an IR absorption table provided in exam to deduce presence or absence of particular bonds or functional groups e.g. C=O 1680 – 1750 cm-1 O-H (acid) 2500- 3000 cm-1 3.6 Analysis Carbon has a value of 12.0000 as it is by definintion the standard reference 1 Mass spectrum for butane 43 29 N Goalby chemrevise.org 2000 1500 Use spectra to identify particular functional groups and to identify impurities, limited to data presented in wavenumber form C=O Spectra for butanal “rogue” absorptions can also occur and are indicators of impurities O-H absorptions tend to be broad Spectra for ethanoic acid C=O The absorption of infra-red radiation by bonds in this type of spectroscopy is the same absorption that bonds in CO2 , methane and water vapour in the atmosphere do that maybe causing global global warming. Absorption or trough in between 1680-1750 cm-1 range indicates presence of C=O bond Absorption or trough in between 2500-3000 cm-1 range indicates presence of O-H bond in an acid Always quote the wave number range from the data sheet Absorption or trough in between 1680- 1750 cm-1 range indicates presence of C=O bond
3.3.6.3 Infrared spectroscopy
Bonds in a molecule absorb infrared radiation at characteristic wavenumbers.
‘Fingerprinting’ allows identification of a molecule by comparison of spectra.
Students should be able to:
• use infrared spectra and the Chemistry Data Sheet or Booklet to identify particular bonds, and therefore functional groups, and also to identify impurities. The link between absorption of infrared radiation by bonds in CO2 , methane and water vapour and global warming.
3.3.6.1 Identification of functional groups by test-tube reactions
The reactions of functional groups listed in the specification.
Students should be able to identify the functional groups using reactions in the specification.



A-level Chemistry exemplar for required practical No. 6 Tests for alcohol, aldehyde, alkene and carboxylic acid: To carry out tests for the presence of organic functional groups and to make accurate observations Student sheet Requirements You are provided with the following: ethanol ethanal or propanal cyclohexene 1-bromobutane dilute ethanoic acid small pieces of metallic sodium under petroleum ether (a beaker of ethanol should be available for safe disposal of any excess sodium) Fehling’s solution A Fehling’s solution B bromine water sodium carbonate solution sodium hydrogencarbonate solid sodium hydroxide solution silver nitrate solution (0.05 mol dm –3 ) dilute nitric acid 250 cm3 beaker anti-bumping granules test tubes, boiling tubes and a test-tube holder thermometer plastic graduated dropping pipettes. Suggested method This experiment is divided into five parts. In every case, you should present all of your observations in a neat table. The presentation of a clearly organised record of your observations is an important skill which you will be expected to demonstrate as part of this assessment. Part 1 – A test for an alcohol a) To about 1 cm 3 of ethanol in a dry test tube, add a small piece of metallic sodium. b) Record your observations. c) Make sure that you dispose safely of any excess sodium using the beaker of ethanol provided. Part 2 – A test for an aldehyde using Fehling’s solution. a) In a clean test tube mix together equal volumes of Fehling’s solution A and Fehling’s solution B. The resultant Fehling’s test reagent should be a clear dark blue solution. b) Add 5 drops of this test reagent to about 1 cm 3 of sodium carbonate solution in a test tube containing a few anti-bumping granules and then add about 1 cm 3 of ethanal (or propanal) to this same test tube. c) Warm the test tube gently for approximately 2 minutes in a beaker half- filled with hot water and then gradually bring the beaker of water to boiling and maintain this temperature for a few minutes. d) Using the test tube holder, carefully lift the test tube out of the boiling water and allow its contents to stand for several minutes. Record your observations. Part 3 – A test for an alkene (a test for unsaturation) a) To about 2 drops of cyclohexene in a test tube, add about 1 cm3 of bromine water and shake the contents of the tube vigorously from side to side. b) Record your observations. Part 4 – A test for a carboxylic acid a) Place one small spatula measure of solid sodium hydrogencarbonate in a boiling tube tube and add to it about 2 cm 3 of dilute ethanoic acid. b) Record your observations. Part 5 – A test for a halogenoalkane a) Using a teat pipette, add 5 drops of 1-bromobutane to about 1 cm3 of sodium hydroxide solution in a test tube. Warm the contents of the test tube for a few minutes, by placing it into a beaker filled with hot water at approximately 60 °C. b) Acidify the contents of the test tube by adding 2 cm 3 of dilute nitric acid and then add about 1 cm 3 of silver nitrate solution. c) Record your observations.
Required practical 6
Tests for alcohol, aldehyde, alkene and carboxylic acid.








