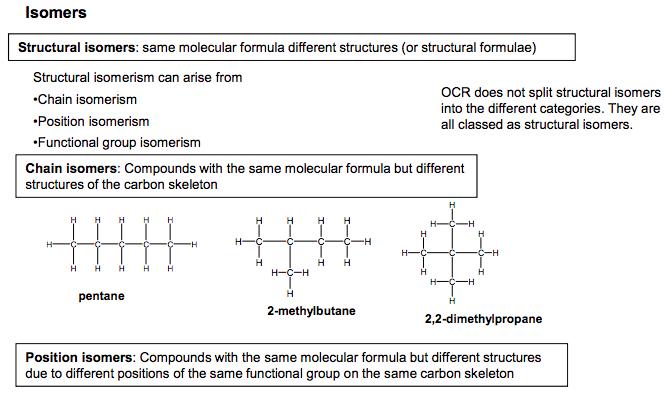
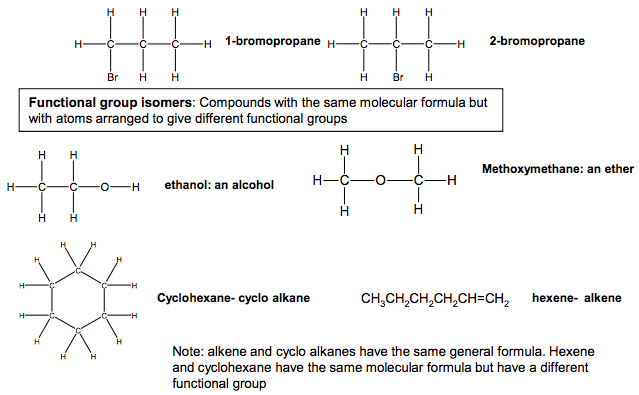
Structural isomers: same molecular formula different structures (or structural formulae) Functional group isomers: Compounds with the same molecular formula but with atoms arranged to give different functional groups Chain isomers: Compounds with the same molecular formula but different structures of the carbon skeleton Isomers Structural isomerism can arise from •Chain isomerism •Position isomerism •Functional group isomerism C C C H H H H H C C H H H H H H H H C C C H H H H H H C C H H H H H H C C C H H H H H C H H C H H H H pentane 2,2-dimethylpropane 2-methylbutane Position isomers: Compounds with the same molecular formula but different structures due to different positions of the same functional group on the same carbon skeleton C C H H Br H C H H H H C C H H H Br C H H H H 1-bromopropane 2-bromopropane C O C H H H H H H C O H H H C H H H ethanol: an alcohol Methoxymethane: an ether C C C C C C H H H H H H H H H H H H Cyclohexane- cyclo alkane CH3CH2CH2CH2CH=CH2 hexene- alkene Note: alkene and cyclo alkanes have the same general formula. Hexene and cyclohexane have the same molecular formula but have a different functional group N Goalby chemrevise.org 5 OCR does not split structural isomers into the different categories. They are all classed as structural isomers.
/
~
~
~
/
4.1.1 Basic concepts of organic chemistry
Isomerism
(e) explanation of the term structural isomers (compounds with the same molecular formula but different structural formulae) and determination of possible structural formulae of an organic molecule, given its molecular formula


 4.1.1 basic concepts and hydrocarbons Page 5
4.1.1 basic concepts and hydrocarbons Page 5 Oxford Textbook Pages : 182 - 183
Oxford Textbook Pages : 182 - 183 CGP Revision Guide Pages : 86 - 87
CGP Revision Guide Pages : 86 - 87