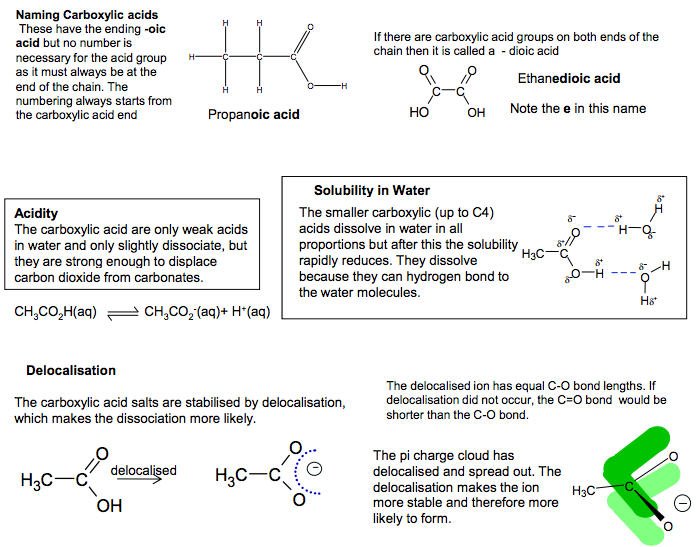


Naming Carboxylic acids These have the ending -oic acid but no number is necessary for the acid group as it must always be at the end of the chain. The numbering always starts from the carboxylic acid end C C O O H H C H H H H Propanoic acid If there are carboxylic acid groups on both ends of the chain then it is called a – dioic acid C C O OH O HO Ethanedioic acid Note the e in this name. Solubility in Water The smaller carboxylic (up to C4) acids dissolve in water in all proportions but after this the solubility rapidly reduces. They dissolve because they can hydrogen bond to the water molecules. H3C C O O H O H H O H H Acidity The carboxylic acid are only weak acids in water and only slightly dissociate, but they are strong enough to displace carbon dioxide from carbonates. CH3CO2H(aq) CH3CO2 -(aq)+ H+ (aq) Delocalisation The carboxylic acid salts are stabilised by delocalisation, which makes the dissociation more likely. delocalised H3C C O OH H3C C O O The delocalised ion has equal C-O bond lengths. If delocalisation did not occur, the C=O bond would be shorter than the C-O bond. H3C C O O The pi charge cloud has delocalised and spread out. The delocalisation makes the ion more stable and therefore more likely to form. Strength of carboxylic acids C C O OH H H Cl CH2 C O OH H3C CH2 C O O delocalised H3C delocalised Increasing chain length pushes electron density on to the COOion, making it more negative and less stable. This make the acid less strong. Alkyl groups electron releasing Chlorine electron withdrawing Electronegative chlorine atoms withdraw electron density from the COOion, making it less negative and more stable. This make the acid more strong. Propanoic acid less acidic than ethanoic acid chloroethanoic acid more acidic than ethanoic. Salt formation reactions of carboxylic acids Carboxylic acids can form salts with metals, alkalis and carbonates. acid + metal (Na) salt + hydrogen 2CH3CO2H + 2Na 2CH3CO2 -Na+ + H2 acid + alkali (NaOH) salt + water CH3CO2H + NaOH CH3CO2 -Na+ + H2O acid + carbonate (Na2CO3 ) salt + water + CO2 2CH3CO2H + Na2CO3 2CH3CO2 -Na+ + H2O + CO2 The effervescence caused by production of CO2 with carboxylic acids with solid Na2CO3 or aqueous NaHCO3 can be used as a functional group test for carboxylic acids. Oxidation of methanoic acid Carboxylic acids cannot be oxidised by using oxidising agents but methanoic acid is an exception as its structure has effectively an aldehyde group C O O H H C O O H + [O] H O It forms carbonic acid (H2 CO3 ) which can decompose to give CO2
/
~
~
~
/
3.3.9.1 Carboxylic acids and esters (A-level only)
The structures of:
• carboxylic acids
Carboxylic acids are weak acids but will liberate CO2 from carbonates.


 3.9 Carboxylic Acids And Derivatives Page 1 - 2
3.9 Carboxylic Acids And Derivatives Page 1 - 2 Oxford Textbook Pages : 415 - 416, 418 - 419
Oxford Textbook Pages : 415 - 416, 418 - 419 CGP Revision Guide Pages : 164
CGP Revision Guide Pages : 164