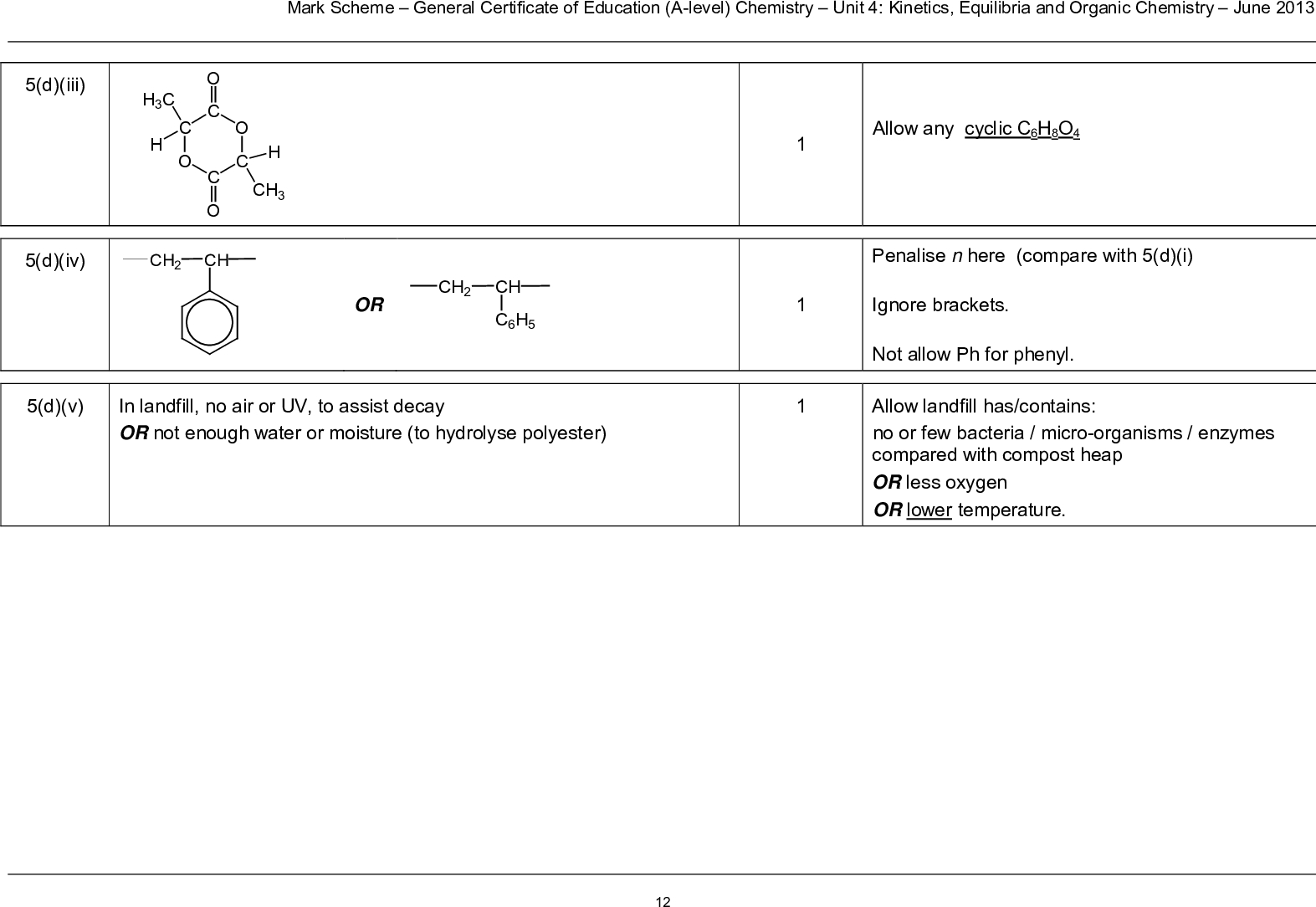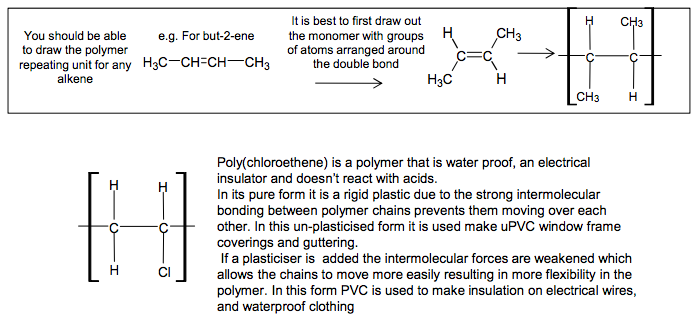
Addition Polymers Addition polymers are formed from alkenes Poly(alkenes) like alkanes are unreactive due to the strong C-C and C-H bonds C C H H CH3 H propene n poly(propene) C C C C C C CH3 H H H CH3 H H H CH3 H H H be able to recognise the repeating unit in a poly(alkene) Poly(propene) is recycled This is called addition polymerisation H3C CH CH CH3 H3C C C CH3 H You should be able H to draw the polymer repeating unit for any alkene It is best to first draw out the monomer with groups of atoms arranged around the double bond e.g. For but-2-ene 4 If asked to draw one repeating unit, don’t add the n on to your diagram, because n represents a large number Add the n’s if writing an equation showing the reaction where ‘n’ monomers become ‘n’ repeating units C C H CH3 H H C C CH3 H H CH3 n N Goalby chemrevise.org C C H CH3 H H C C H Cl H H Poly(chloroethene) is a polymer that is water proof, an electrical insulator and doesn’t react with acids. In its pure form it is a rigid plastic due to the strong intermolecular bonding between polymer chains prevents them moving over each other. In this un-plasticised form it is used make uPVC window frame coverings and guttering. If a plasticiser is added the intermolecular forces are weakened which allows the chains to move more easily resulting in more flexibility in the polymer. In this form PVC is used to make insulation on electrical wires, and waterproof clothing
3.3.4.3 Addition polymers
Addition polymers are formed from alkenes and substituted alkenes.
The repeating unit of addition polymers.
IUPAC rules for naming addition polymers.
Addition polymers are unreactive.
Appreciate that knowledge and understanding of the production and properties of polymers has developed over time.
Typical uses of poly(chloroethene), commonly known as PVC, and how its properties can be modified using a plasticiser.
Students should be able to:
• draw the repeating unit from a monomer structure
• draw the repeating unit from a section of the polymer chain
• draw the structure of the monomer from a section of the polymer
• explain why addition polymers are unreactive
• explain the nature of intermolecular forces between molecules of polyalkenes.




 3.4 Alkenes Page 4
3.4 Alkenes Page 4 Oxford Textbook Pages : 224 - 227
Oxford Textbook Pages : 224 - 227 CGP Revision Guide Pages : 146 - 147
CGP Revision Guide Pages : 146 - 147