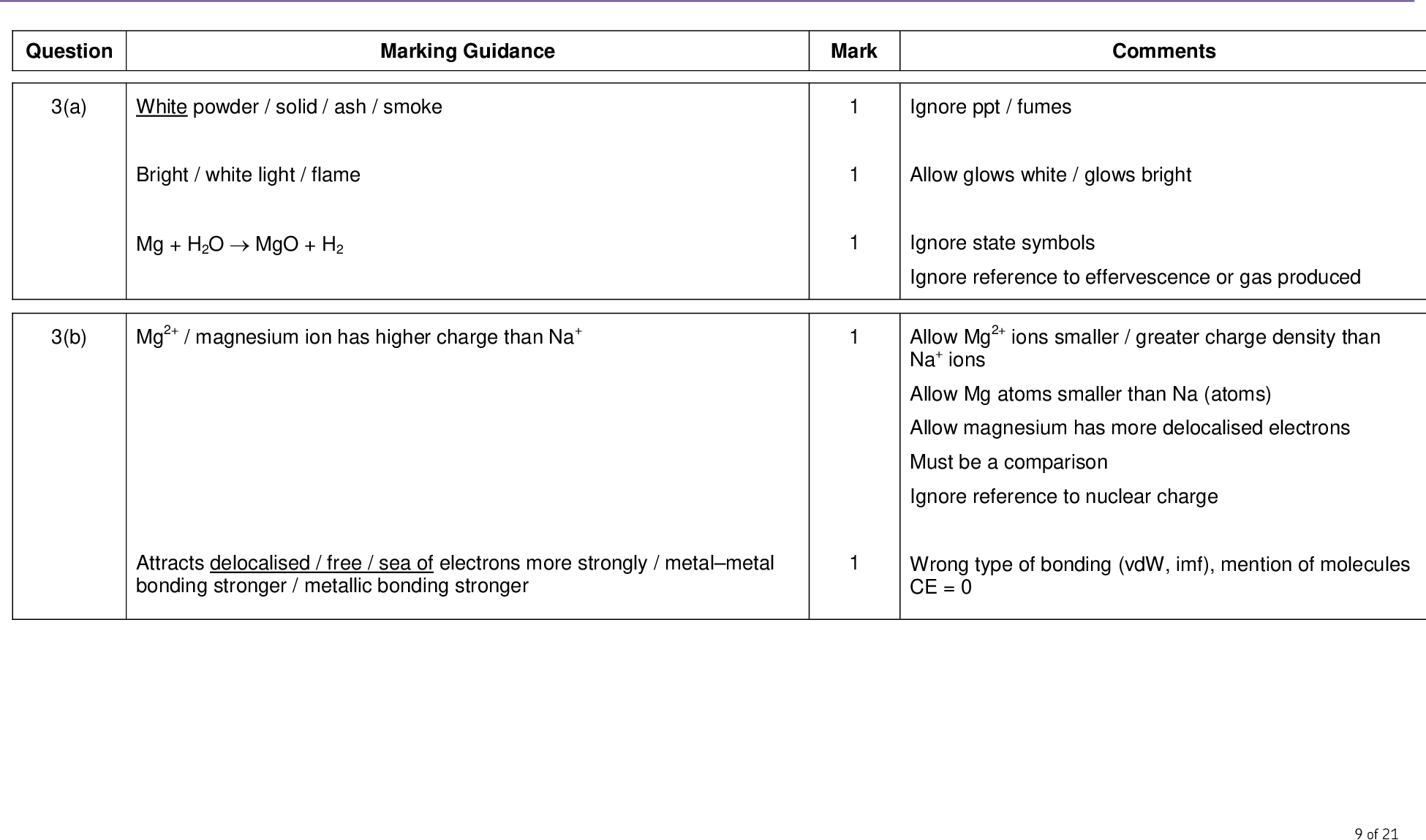
Trends in the reactions of the elements with water, limited to Na and Mg Sodium reacts with cold water. It fizzes around on surface etc. 2 Na (s) + 2 H2O (l) 2 NaOH (aq) + H2 (g) Learn the equations Magnesium reacts very slowly with cold water to form the hydroxide but reacts more readily with steam to form the oxide Mg (s) + H2O (g) MgO (s) + H2 (g) Trends in the reactions of the elements Na, Mg, Al, Si, P and S with oxygen The elements all react with oxygen to form oxides. Sodium burns with a yellow flame to produce a white solid Mg, Al, Si and P burn with a white flame to give white solid smoke S burns with a blue flame to form an acidic choking gas. 4 Na (s) + O2 (g) 2 Na2O (s) 2Mg (s) + O2 (g) 2MgO (s) 4Al + 3O2 (g) 2Al2O3 (s) Si + O2 (g) SiO2 (s) 4P + 5O2 (g) P4O10 (s) S+ O2 (g) SO2 (g) You should be able to write these equations. Learn the formulae of the oxides Sodium is stored under oil and phosphorus under water to stop these elements coming into contact and reacting with air
/
~
~
~
/
3.2.4 Properties of Period 3 elements and their oxides (A-level only)
The reactions of Na and Mg with water.
The trends in the reactions of the elements Na, Mg, Al, Si, P and S with oxygen, limited to the formation of Na2O, MgO, Al2O3 , SiO2 , P4O10, SO2 and SO3


 2.4 Period 3 Page 1
2.4 Period 3 Page 1 Oxford Textbook Pages : 346 - 348
Oxford Textbook Pages : 346 - 348 CGP Revision Guide Pages : 106
CGP Revision Guide Pages : 106