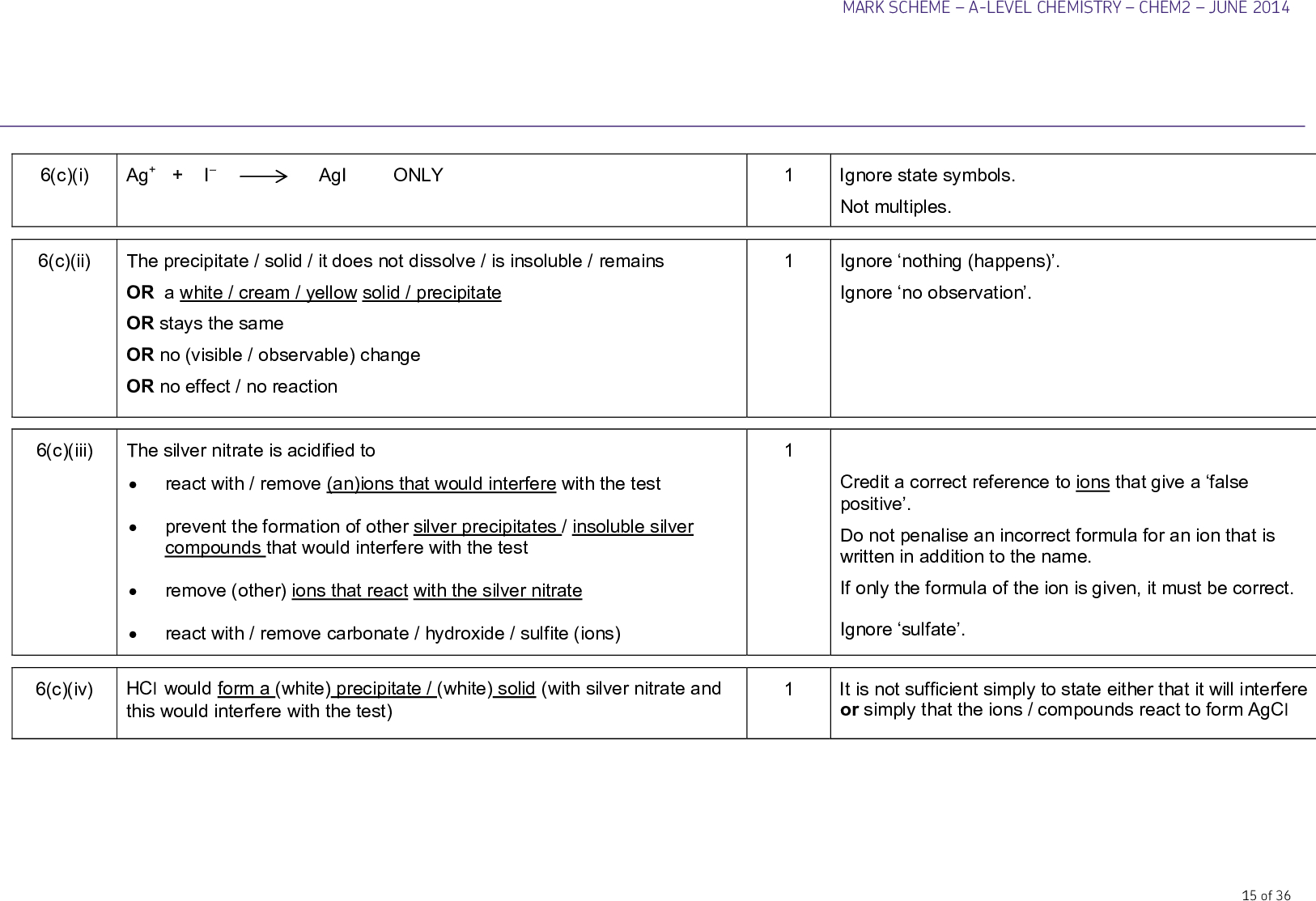
2. The reactions of halide ions with silver nitrate. This reaction is used as a test to identify which halide ion is present. The test solution is made acidic with nitric acid, and then Silver nitrate solution is added dropwise. The role of nitric acid is to react with any carbonates present to prevent formation of the precipitate Ag2CO3 . This would mask the desired observations 2 HNO3 + Na2CO3 2 NaNO3 + H2O + CO2 Fluorides produce no precipitate Chlorides produce a white precipitate Ag+ (aq) + Cl- (aq) AgCl(s) Bromides produce a cream precipitate Ag+ (aq) + Br- (aq) AgBr(s) Iodides produce a pale yellow precipitate Ag+ (aq) + I- (aq) AgI(s) The silver halide precipitates can be treated with ammonia solution to help differentiate between them if the colours look similar: Silver chloride dissolves in dilute ammonia to form a complex ion AgCl(s) + 2NH3 (aq) [Ag(NH3 )2 ] + (aq) + Cl- (aq) Colourless solution Silver bromide dissolves in concentrated ammonia to form a complex ion AgBr(s) + 2NH3 (aq) [Ag(NH3 )2 ] + (aq) + Br – (aq) Colourless solution Silver iodide does not react with ammonia – it is too insoluble.
/
~
~
~
/
3.2.3.1 Trends in properties
The trend in reducing ability of the halide ions, including the reactions of solid sodium halides with concentrated sulfuric acid. The use of acidified silver nitrate solution to identify and distinguish between halide ions. The trend in solubility of the silver halides in ammonia.
Students should be able to explain why:
• silver nitrate solution is used to identify halide ions
• the silver nitrate solution is acidified
• ammonia solution is added.


 2.3 Halogens Page 1
2.3 Halogens Page 1 Oxford Textbook Pages : 165 - 166
Oxford Textbook Pages : 165 - 166 CGP Revision Guide Pages : 103
CGP Revision Guide Pages : 103